Product Overview
Background
Reduced glutathione (GSH) is a tripeptide (γ-glutamylcysteinylglycine) that contains a free thiol group. GSH is a major tissue antioxidant that provides reducing equivalents for the glutathione peroxidase (GPx) catalyzed reduction of lipid hydroperoxides to their corresponding alcohols and hydrogen peroxide to water.
In the GPx catalyzed reaction, the formation of a disulfide bond between two GSH molecules gives rise to oxidized glutathione (GSSG). The enzyme glutathione reductase (GR) recycles GSSG to GSH with the simultaneous oxidation of β-nicotinamide adenine dinucleotide phosphate (β-NADPH2).
When cells are exposed to increased levels of oxidative stress, GSSG will accumulate and the ratio of GSH to GSSG will decrease. Therefore, the determination of the GSH/GSSG ratio and the quantification of GSSG are useful indicators of oxidative stress in cells and tissues.
Assay Principle
The low concentration of GSSG (high GSH/GSSG ratio) in tissues coupled with the need to prevent GSH oxidation during sample preparation, are important considerations for the accurate measurement of GSSG and GSH/GSSG ratios. Guntherberg and Rost (2) first reported the use of N ethylmaleimide (NEM) reacting with GSH to form a stable complex, therefore removing the GSH prior to the quantification of GSSG in tissues. Unfortunately, NEM inhibits GR. To overcome this problem, Griffith (3) employed 2 vinylpyridine (2-VP) to derivatize GSH. Although 2-VP does not significantly inhibit GR, it reacts relatively slowly with GSH and is not very soluble in aqueous solutions.
Scavenging of Free Thiols: This assay employs a pyridine derivative as a thiol-scavenging reagent thereby overcoming the shortfalls of both prior methods. At the concentration employed in the assay, this derivative reacts quickly with GSH but does not interfere with the GR activity.
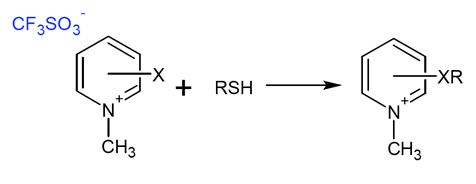
Figure 1
: Reaction of the scavanger with a thiol to form a pyridinium salt
Thiol Quantification: The quantitative determination of the total amount of glutathione (GSH + GSSG) employs the enzymatic method first reported by Tietze (1). Briefly, the reaction of GSH with Ellman’s reagent (5,5’-dithiobis-2-nitrobenzoic acid (DTNB)) gives rise to a product that can be quantified spectrophotometrically at 412 nm. This reaction is used to measure the reduction of GSSG to GSH. The rate of the reaction is proportional to the GSH and GSSG concentration.
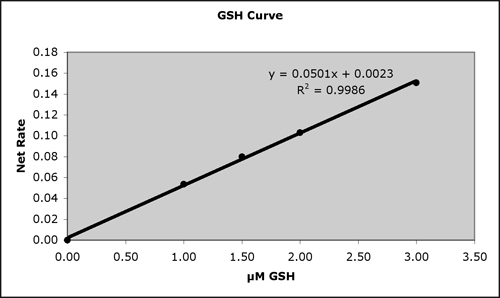
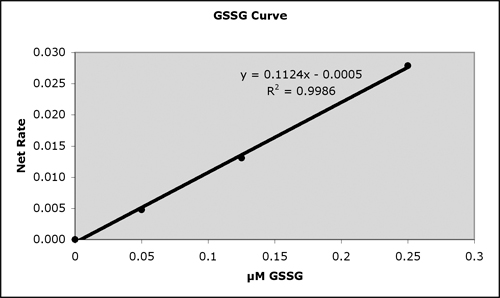
Typical Standard Curves
References
1. Tietze, F., (1969) Analytical Chemistry 27, 502-520.
2. Guntherberg, H. and Rost, J., (1966) Analytical Biochemistry 15, 205-210.
3. Griffith, O.W., (1980) Analytical Biochemistry 106, 207-212.
4. Richie, J.P. Jr., et al, (1996) Clinical Chemistry 42, 64-70.
5. Anderson, M., (1996) Glutathione in Free Radicals, A Practical Approach, 213.